EMH Schweizerischer Ärzteverlag AG
Farnsburgerstrasse 8
CH-4132 Muttenz
+41 (0)61 467 85 44
support@swisshealthweb.ch
www.swisshealthweb.ch
Published on 26.02.2019
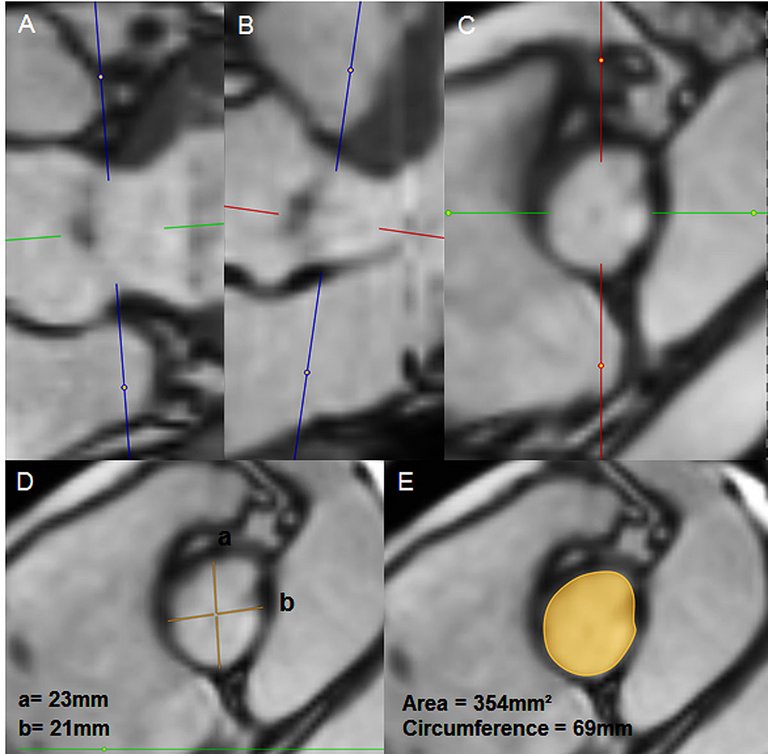
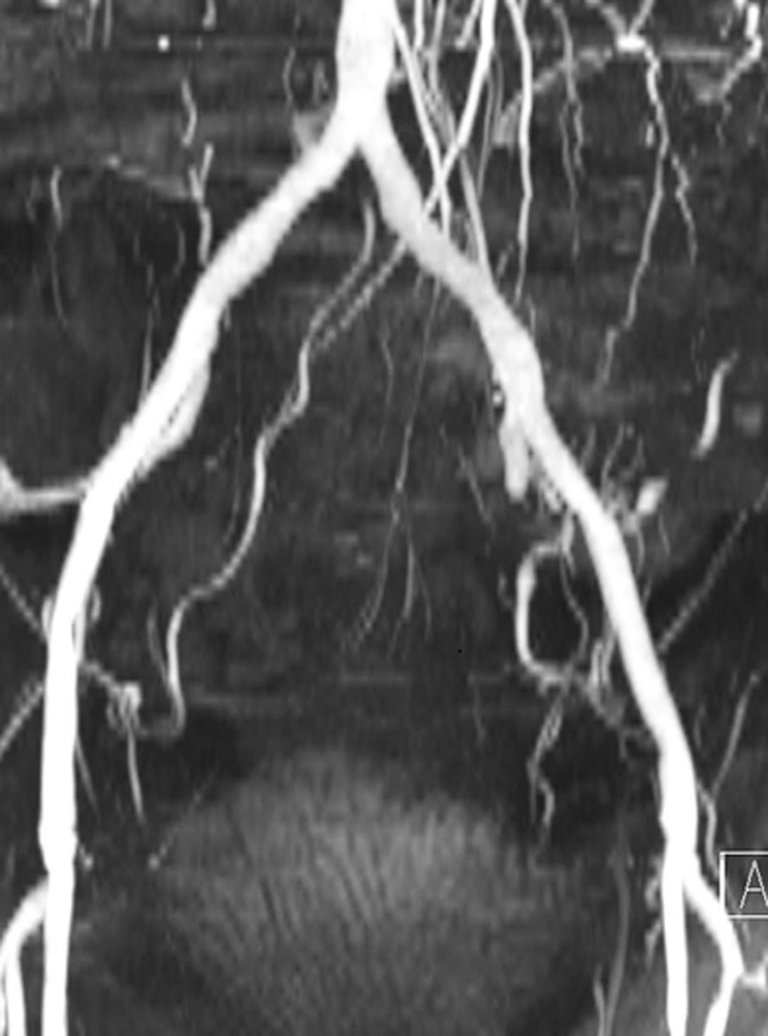
Gadolinium-free MR can be used for pre-procedural annular and iliofemoral assessment, and thus selection of prosthesis size.
| Magnet resonance tomography without gadolinium instead of multislice computed tomography |
| Coronary angiography with 3-4 views |
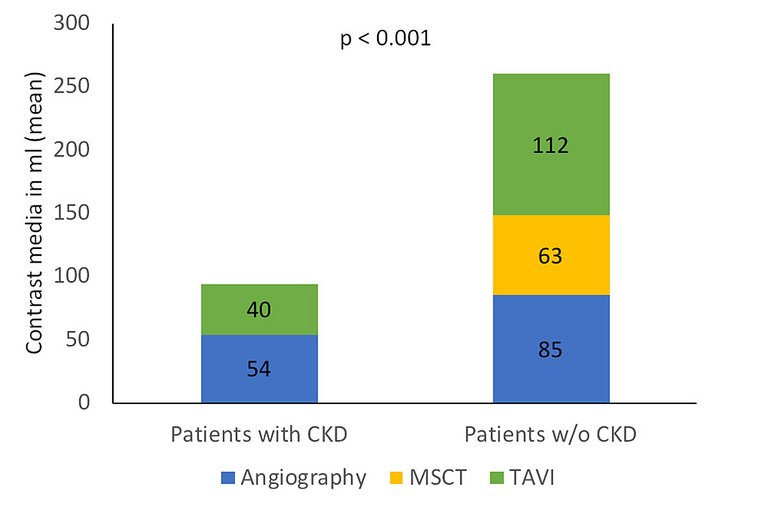
| Prehydration with 500 ml of saline 0.9% |
| Transcatheter aortic valve replacement under conscious sedation |
| Rapid pacing for determination of implant view |
| Transthoracic echocardiography to assess paravalvular regurgitation |
| Careful postprocedural monitoring and care |
| All patients (n = 168) | Patients with severe CKD (n = 20) | Patients without severe CKD (n = 148) | p value | ||
|---|---|---|---|---|---|
| Age, years | 81±6 | 84±6 | 81±6 | 0.05 | |
| Female sex | 92 (55%) | 13 (65%) | 79 (53%) | 0.35 | |
| Hypertension | 142 (85%) | 17 (85%) | 125 (84%) | 1.0 | |
| Diabetes | 30 (18%) | 4 (20%) | 26 (18%) | 0.76 | |
| Coronary artery disease | 83 (49%) | 9 (45%) | 74 (50%) | 0.81 | |
| Prior stroke | 19 (11%) | 1 (5%) | 18 (12%) | 0.48 | |
| STS PROM, % | 4.3±3.7 | 8.2±4.0 | 3.8±3.4 | <0.01 | |
| Echocardiographic parameters | Aortic valve area, cm2 | 0.76±0.19 | 0.7±0.22 | 0.77±0.19 | 0.16 |
| Mean gradient, mmHg | 47±17 | 45±13 | 48±18 | 0.52 | |
| Left ventricular ejection fraction, % | 59±13 | 51±15 | 58±13 | 0.03 | |
| Laboratory parameters | Creatinine (µmol/L) | 107±56 | 198±62 | 94±42 | <0.01 |
| GFR ( ml/min) | 54±24 | 24±5 | 58±23 | <0.01 | |
| Implanted valve | Acurate neo | 106 (63%) | 14 (70%) | 92 (62%) | 0.95 |
| Sapien 3 | 49 (29%) | 5 (25%) | 44 (30%) | ||
| Evolut R | 10 (6%) | 1 (5%) | 9 (7%) | ||
| Allegra | 3 (2%) | 0 (0%) | 3 (2%) | ||
| All patients (n = 168) | Patients with severe CKD (n = 20) | Patients without severe CKD (n = 148) | p value | ||
| Aortic valve area, cm2 | 2.1±0.5 | 2.1±0.5 | 2.1±0.5 | 0.64 | |
| Mean gradient, mmHg | 6.8±3.6 | 5.5±3.3 | 6.9±3.7 | 0.12 | |
| Paravalvular regurgitation | none/trace | 103 (64%) | 12 (60%) | 91 (64%) | 0.85 |
| mild | 57 (35%) | 8 (40%) | 49 (35%) | ||
| moderate | 2 (1.2%) | 0 (0%) | 2 (1.4%) | ||
| Acute kidney injury stage 2 or 3 | 5 (3.0%) | 1 (5.0%) | 4 (2.7%) | 0.054 | |
| All patients (n = 168) | Patients with severe CKD (n = 20) | Patients without severe CKD (n = 148) | p value | |
|---|---|---|---|---|
| All-cause mortality | 3 (1.8%) | 0 (0%) | 3 (2.0%) | 0.68 |
| Device success | 159 (96.4%) | 20 (100%) | 139 (95.9%) | 1.0 |
| Early safety | 150 (91%) | 18 (90%) | 132 (91%) | 1.0 |
| Clinical efficacy | 156 (95.1%) | 20 (100%) | 136 (94.4%) | 0.6 |
| Any stroke | 0 (0%) | 0 (0%) | 0 (0%) | N/A |
| New permanent pacemaker | 6 (3.6%) | 2 (10%) | 4 (2.7%) | 0.15 |
| Major vascular complication | 11 (6.6%) | 1 (5%) | 10 (6.8%) | 1.0 |
| Major or life-threatening bleeding | 12 (7.1%) | 1 (5%) | 11 (7.4%) | 1.0 |
Published under the copyright license.
"Attribution - Non-Commercial - NoDerivatives 4.0"
No commercial reuse without permission.
See: emh.ch/en/emh/rights-and-licences/