EMH Schweizerischer Ärzteverlag AG
Farnsburgerstrasse 8
CH-4132 Muttenz
+41 (0)61 467 85 44
support@swisshealthweb.ch
www.swisshealthweb.ch
Published on 12.03.2019
| Year | First author [ref.] | Number of patients | Male, n (%) | 3D echo algorithm | 3D echo device |
|---|---|---|---|---|---|
| 2004 | Kühl P, et al. [10] | 24 | n/a | Semiautomated | Sonos 7500, Philips |
| 2008 | Pouleur AC, et al. [11] | 83 | 67 (80.7%) | Semiautomated | IE33 Philips |
| 2008 | Mor-Avi V, et al. | 92 | 69 (75%) | Semiautomated | IE33 Philips |
| 2012 | Thavendiranathan P, et al. [12] | 91 | 19 (20.8%); 23 (25.2%); 17 (18.6%) | Automated | Acuson SC2000, Siemens |
| 2012 | Moceri P, et al. [13] | 24 | 17 (70.8%) | Semiautomated | IE33 Philips |
| 2012 | Shibayama K, et al. [14] | 41 | 30 (73.1%) | Automated | Acuson SC2000, Siemens |
| 2014 | Aurich M, et al. | 47 | 21 (44.6%) | Automated | Vivid E9 BT 11, GE |
| 2014 | Driessen M, et al. [15] | 35 | 19 (54%) | Semiautomated | IE33 Philips; Artida-4D, Toshiba |
| 2016 | Tsang W, et al. [16] | 159 | 44 (27.6%); 31 (19.4%) | Automated | EPIQ and IE33, Philips |
| 2016 | Levy F, et al. [17] | 54 | 40 (74%) | Automated | EPIQ 7C, Philips |
| 2017 | Tamborini G, et al. [18] | 189 | 130 (68.7%) | Semiautomated | EPIQ and IE33, Philips |
| 2018 | Muraru D, et al. [19] | 92 | 63 (68.5%) | Automated | Acuson SC2000, Siemens; IE33 Philips; Vivid E9 BT 11, GE |
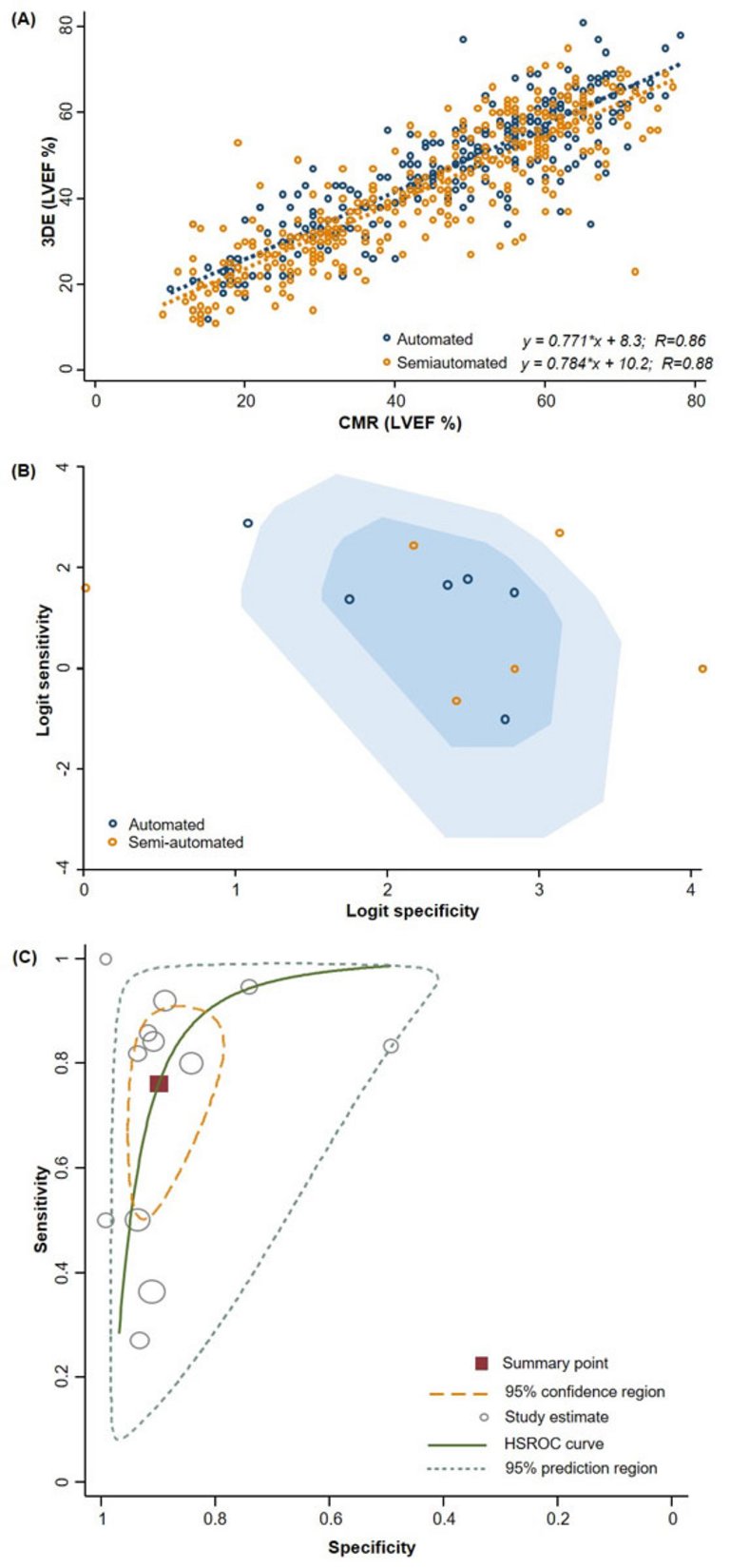
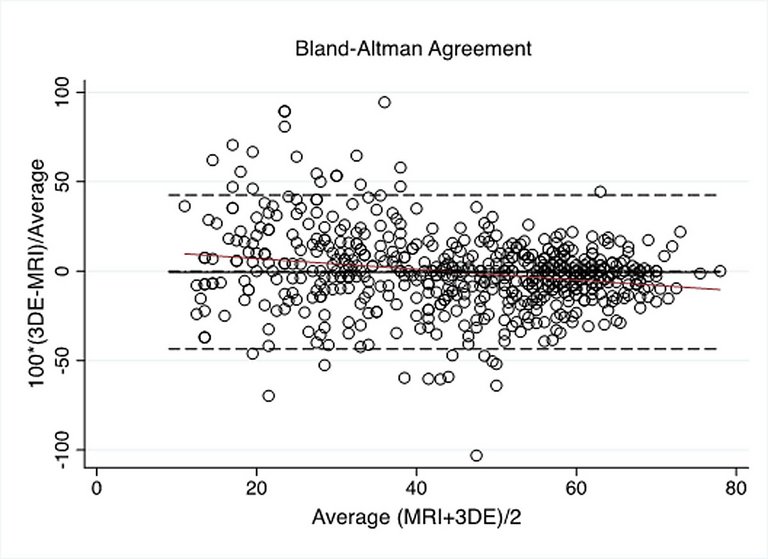
Published under the copyright license.
"Attribution - Non-Commercial - NoDerivatives 4.0"
No commercial reuse without permission.
See: emh.ch/en/emh/rights-and-licences/