EMH Schweizerischer Ärzteverlag AG
Farnsburgerstrasse 8
CH-4132 Muttenz
+41 (0)61 467 85 44
support@swisshealthweb.ch
www.swisshealthweb.ch
Published on 17.12.2019
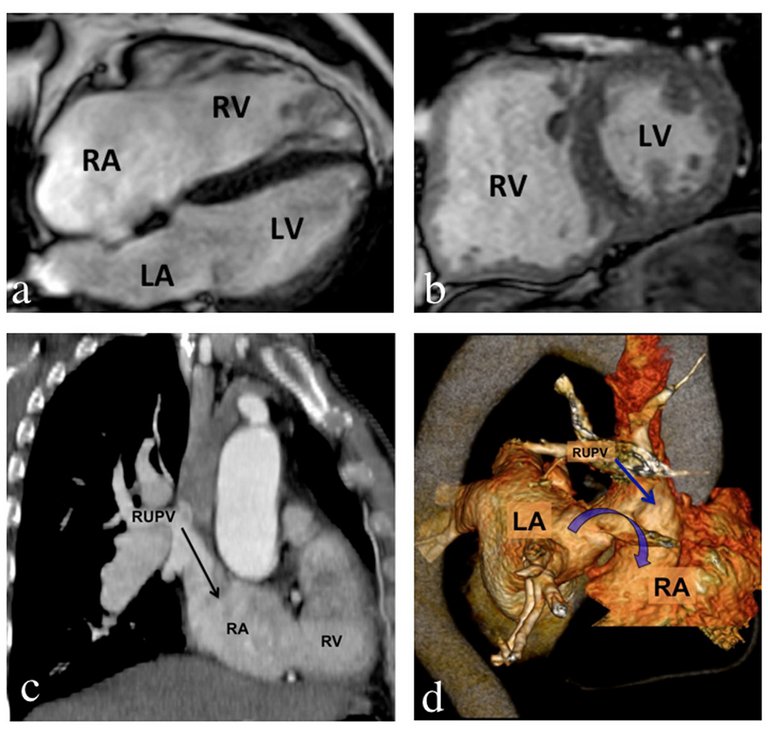
Interatrial communications are the most frequent congenital heart malformation in adulthood. We report typical examples of sinus venosus and unroofed coronary sinus defects associated with anomalous pulmonary venous return.
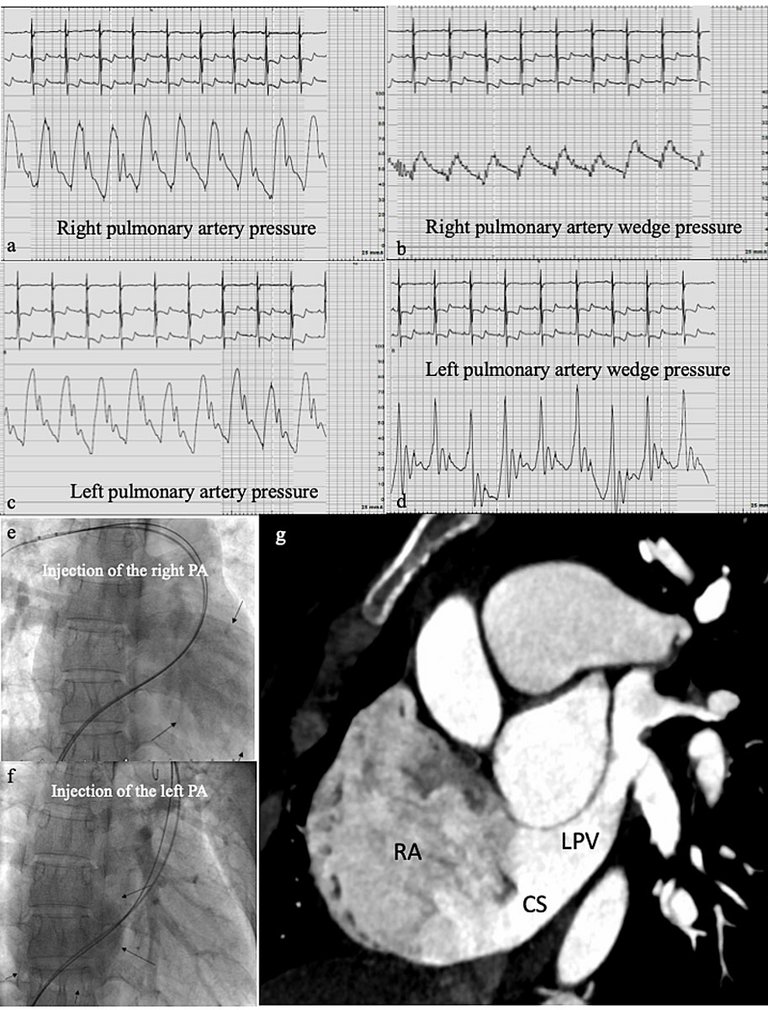
| Baseline | With oxygen | With nitric oxide | |
|---|---|---|---|
| Systemic pressure (mm Hg; s/d-m) | 107/60-83 | – | – |
| Systolic PAP (mm Hg) | Right: 82 Left: 76 | 72 | 72 |
| Diastolic PAP (mm Hg) | Right: 39 Left: 36 | 27 | 30 |
| Mean PAP (mm Hg) | Right: 57 Left: 52 | 48* | 49* |
| Wedge (mm Hg) | Right: 21 Left: 10 | – | – |
| LVEDP (mm Hg) | 15 | – | – |
| Qp/Qs | 1.63 | – | – |
| Systemic cardiac output (l/min) | 3.74 | – | – |
| Pulmonary cardiac output (l/min) | 6.10 | – | – |
| TPG | 36 | – | – |
| PVR (WU) | (57-21)/6.1=5.9 | – | – |
| SVR (WU) | (83-2)/3.74= 21.6 | – | – |
Published under the copyright license.
"Attribution - Non-Commercial - NoDerivatives 4.0"
No commercial reuse without permission.
See: emh.ch/en/emh/rights-and-licences/