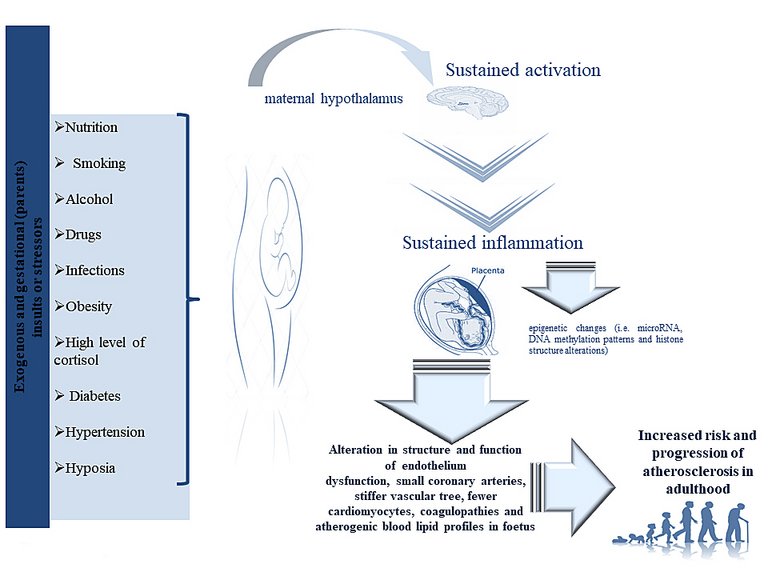
Given the well-recognised role of inflammation in the onset of CVDs such as atherosclerosis and CHDs, its importance in offspring that have experienced prenatal inflammation exposure (PIE) has also been investigated. Specifically, it has been demonstrated that maternal influenza exposure and febrile genitourinary infections affect fetal cardiovascular development, predisposing the progeny to CVDs such as atherosclerosis [
32–
36]. Similar data have been obtained by investigating maternal pre-pregnancy obesity and diabetes, and excessive gestational weight gain. Specifically, under these maternal conditions an increased activation of inflammatory pathways and a sustained macrophage infiltration in the placenta has been detected [
37–
40]. Furthermore, early onset of maternal higher blood pressure or preeclampsia during pregnancy have been associated with a sustained response of inflammatory CD4
+ T cell subsets, accompanied by higher plasma levels of interleukin-6 (IL-6), IL-17 and tumour necrosis factor (TNF)-α in mothers [
41]. In addition, it has been demonstrated that maternal hypertension can affect early childhood blood pressures and cardiovascular health in the offspring [
42,
43]. Maternal smoking has been also demonstrated to cause a proinflammatory state, including elevated maternal serum levels of TNF-α and IL-1β, that is recognised as an independent risk factor for CVDs [
44–
46]. This sustained inflammation has been also demonstrated to be the result of the crosstalk among inflammatory pathways (such as TLR-4), oxidative stress, over-activation of the renin-angiotensin system (RAS), NF-κB (nuclear factor “kappa-light-chain-enhancer” of activated B cells) deregulated homeostasis, epigenetic reprograming, and dysregulation of the immune system and the hypothalamic-pituitary-adrenal axis [
47]. This evidence has been recently stressed by Deng and co-workers in a well-structured review [
47]. Moreover, oral administration with pyrrolidine dithio-carbamate (PDTC), a potent antioxidant, in the second pregnancy trimester has been shown to effectively prevent PIE-programmed hypertension, vascular damage [
48] and myocardial remodelling [
49] in rats. In addition, early postnatal treatment with PDTC, N-acetyl-l-cysteine (NAC), a glutathione precursor, or Tempol, a superoxide dismutase-mimetic drug, has been found to prevent PIE-programmed CVDs in animal models [
48]. Consequently, treatments against inflammation, oxidative stress, etc. in prenatal and early postnatal life might be appropriate therapeutic methods for the prevention of PIE-CVDs.