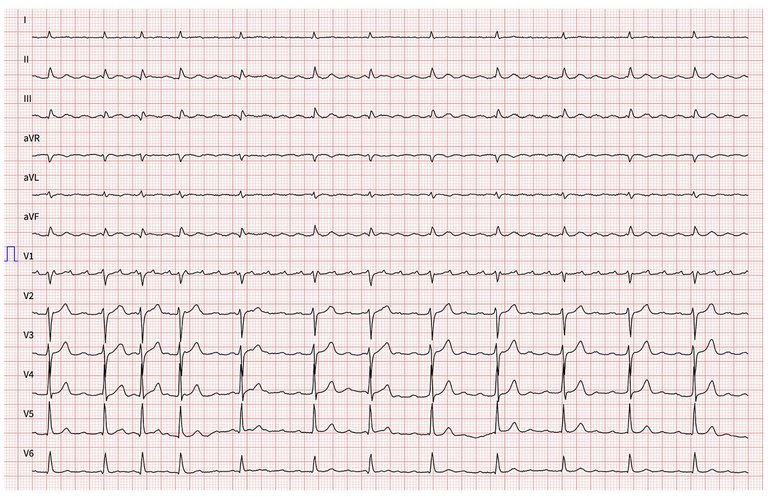
Continuous monitoring of the surface 12-lead ECG and bipolar endocardial electrograms during mapping and ablation were stored on a computer-based digital amplifier/recorder system (Bard Boston Scientific, Inc., Marlborough). Intracardiac electrograms were filtered from 30 to 500 Hz and measured at a sweep speed of 100 mm/s. A standard decapolar catheter was placed in the coronary sinus and served as the reference electrode. The eccentric coronary sinus activation (with the coronary sinus catheter in a proximal position) during atrial tachycardia suggested a left atrial origin. A quadripolar electrode was placed in the His position for baseline measurements, as well as for guiding transseptal puncture. The 12-lead surface ECG during narrow complex tachycardia constituting an atrial tachycardia with variable atrioventricular conduction showed a positive P-wave in V1 and positive P-waves in the precordial leads, as well as an inferior P wave axis, suggesting an origin in the superior part of the left atrium. The tachycardia was stable with an atrial cycle length of 270 ms (
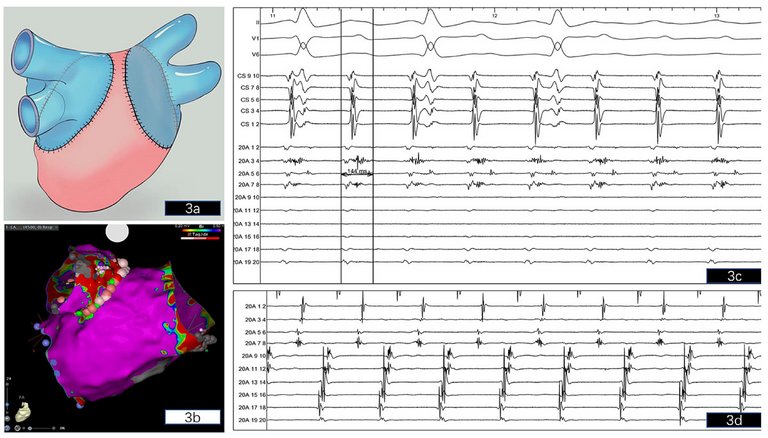
fig. 2). Single transseptal access was obtained under fluoroscopy using a 8.5 F SL-1 sheath and a BRK-1 (Abbott, Chicago, Il) transseptal needle. Next, the SL1 sheath was replaced by an Agilis steerable sheath (8.5 F, Abbott) and a multipolar high-resolution mapping catheter (PentaRay, Biosense Webster, Diamond Bar, CA) and later a 3.5-mm-tip SmartTouch ThermoCool (Biosense Webster) bidirectional ablation catheter were placed in the left atrium. A 3D endocardial high-density (Confidense module, Biosense Webster) electroanatomical activation and voltage map of the left atrium during atrial tachycardia was acquired with the PentaRay and ablation catheter covering the recipient’s left atrium and the donor pulmonary veins guided by contrast-enhanced computed tomography (CT) of the left atrium that was acquired one day prior to the procedure to exclude left atrial thrombi and to reconstruct 3D left atrial anatomy. The CT and 3D bipolar endocardial voltage mapping during atrial tachycardia confirmed that the donor bilateral pulmonary veins shared an ipsilateral common trunk (single right-sided pulmonary vein and single left-sided pulmonary vein) that was created by the thoracic surgeon during bilateral lung transplantation in order to facilitate the anastomosis of the pulmonary veins with the left atrium of the recipient. A schematic view of the donor pulmonary vein anastomosis during bilateral lung transplantation shows ipsilateral donor pulmonary veins (coloured blue) taken as common trunks with a cuff of left atrial tissue for anastomosis to the recipient left atrium (coloured pink) (
fig. 3a). Theoretically, this results in complete electrical isolation of the donor pulmonary veins / atrial cuff. However, during voltage and activation mapping, the suture lines and pulmonary vein cuffs manifested as low-voltage areas, whereas the left atrium displayed no larger low voltage areas (
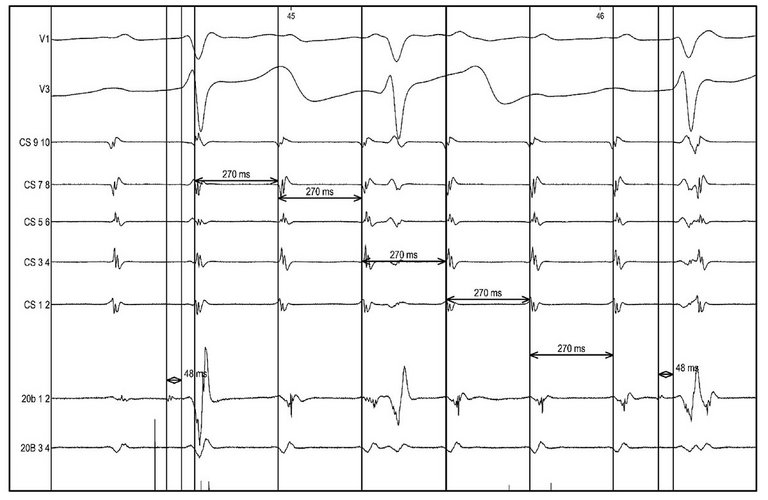
fig. 3b). The right common trunk pulmonary vein was surgically isolated. However, positioning the PentaRay catheter across the suture line between the common trunk left pulmonary vein and left atrium recorded long fractionated low-voltage potentials and separated atrial electrograms, located at the posterosuperior and anterosuperior aspects of the left pulmonary vein-left atrium anastomosis (
fig. 3c, d), indicating viable myocytes and a partial line of block in this area. The activation map confirmed a macroreentrant atrial tachycardia (donor-to-recipient conduction) involving the left donor pulmonary vein with an exit at the posterosuperior aspect of the pulmonary vein-LA junction displaying long fractionated low-voltage potentials (
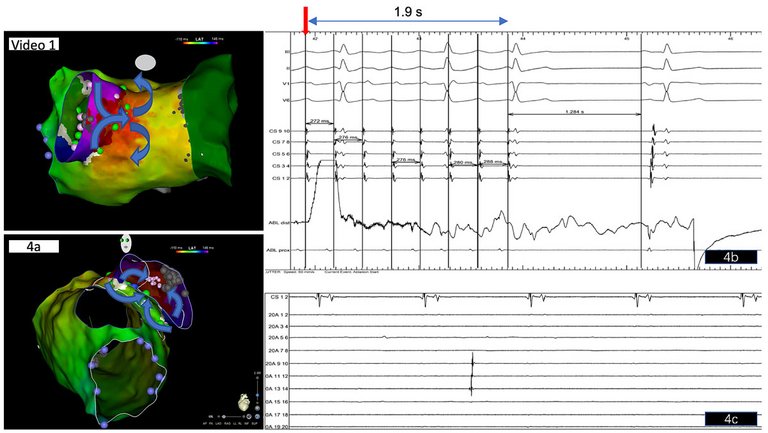
fig. 4a, video 1). Based on our findings, the critical isthmus of the reentrant circuit and exit of the atrial tachycardia was suspected at the posterosuperior aspect of the left pulmonary vein-LA anastomosis displaying the longest fractionated bipolar signals (
fig. 3c; 20A 3-4). As previously published by other groups [
3,
4], the conduction gap along this anastomosis line acted as the critical isthmus of the reentry circuit, and the electrical propagation wave-front entered the donor pulmonary vein cuff at a second gap at the anterior ridge between the recipient left atrium and donor pulmonary vein, closing the circuit (
fig. 4a). Radiofrequency ablation in this area terminated the tachycardia within 2 s with slowing down of the atrial tachycardia prior to termination (
fig 4 b). Since a macroreentrant tachycardia incorporating the left pulmonary vein was assumed, based on 3D activation mapping, we opted for electrical isolation of the left pulmonary vein common trunk (
fig. 3b and video1), which was achieved at the end of the procedure after a waiting period of 30 min, showing isolated pulmonary vein activity with exit block in this vein (
fig. 4c).